Predicting Unseen Gene Perturbation Response Using Graph Neural Networks with Biological Priors
Predicting Unseen Gene Perturbation Response Using Graph Neural Networks with Biological Priors
Dip, S. A.; Zhang, L.
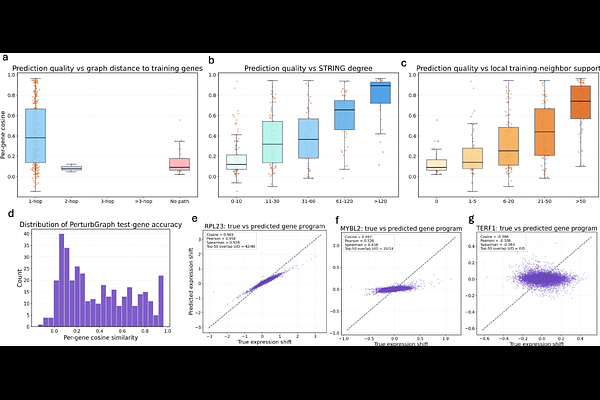
AbstractPredicting transcriptional responses to genetic perturbations is a central challenge in functional genomics. CRISPR Perturb-seq experiments measure gene expression changes induced by targeted perturbations, yet experimentally testing all possible perturbations remains infeasible. Computational models that infer responses for unseen perturbations are therefore essential for scalable functional discovery. We introduce PerturbGraph, a biologically informed graph-learning framework for predicting transcriptional responses of unseen gene perturbations by integrating interaction networks, functional annotations, and transcriptional features. Our approach is motivated by the observation that perturbation effects propagate through molecular interaction networks and manifest as coordinated transcriptional programs. Starting from single-cell CRISPR perturbation data, we construct perturbation signatures representing expression shifts relative to control cells and project them into a compact latent program space that captures stable transcriptional variation while reducing noise. Each gene is represented using enriched biological features integrating protein-protein interaction network embeddings, network topology statistics, baseline transcriptional characteristics, and Gene Ontology annotations. A graph neural network propagates information across the interaction network to infer perturbation programs for genes whose effects are not observed during training. Across unseen-perturbation benchmarks, PerturbGraph consistently outperforms classical machine learning models, perturbation-specific deep learning approaches such as scGen and CPA, and alternative graph neural architectures. The model achieves up to 6% improvement in cosine similarity over strong tree-based baselines and more than 20% improvement over linear models while improving recovery of differentially expressed genes. These results show that integrating biological interaction networks with graph representation learning enables accurate prediction of transcriptional effects for previously unobserved genetic perturbations. Code is publicly available at https://github.com/Sajib-006/PerturbGraph.