Transcription Factor Subtype Governs Response and Resistance to DLL3-Directed T-Cell Engagement in Small Cell Lung Cancer
Transcription Factor Subtype Governs Response and Resistance to DLL3-Directed T-Cell Engagement in Small Cell Lung Cancer
Vasseur, D.; Saito, S.; Gulati, G. S.; Lee, G. G.; Laimon, Y. N.; Simsek, B.; Lerner, M.; Cho, H.; Li, Y.; Wang, T.; Seo, J.-H.; Savignano, H.; James, B.; Zhang, Z.; Semaan, K.; Jin, Z.; Daoud Khatoun, W.; Nafeh, G.; Nawfal, R.; Cooper, A. J.; Miller, K.; Seager, M. D.; Brea, E. J.; Smith, E.; Chang, J.; Pelletier, M.; Costa, C.; Choueiri, T. K.; Signoretti, S.; Sands, J.; Baca, S. C.; Freedman, M. L.; Oser, M. G.
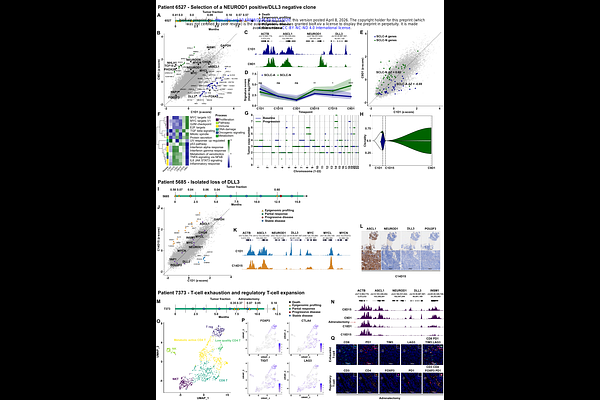
AbstractAlthough small cell lung cancer (SCLC) comprises transcription factor (TF)-defined molecular subtypes (ASCL1, NEUROD1, POU2F3), the extent to which these subtypes predict response to clinically effective therapy in patients-and whether therapy can select for subtype switching-remains unknown. The recent approval of the DLL3 x CD3 bispecific T-cell engager tarlatamab represents one of the first meaningful advances in relapsed small cell lung cancer (SCLC) in decades, yet responses remain heterogeneous and resistance is inevitable. Here, we inferred SCLC gene expression from circulating chromatin in prospectively collected patient plasma (46 patients; 167 samples), enabling interrogation of response and acquired resistance to tarlatamab. Parallel development of the first immunocompetent syngeneic mouse model to study tarlatamab response and resistance enabled functional validation. Across species, findings converged on a central principle: TF subtype governs both initial response and acquired resistance. Therapeutic response was significantly associated with ASCL1-subtype tumors, whereas NEUROD1-subtype tumors exhibited inferior responses and POU2F3-subtype tumors were uniformly resistant, consistent with DLL3 being a direct ASCL1 transcriptional target and most highly expressed in ASCL1-positive tumors. Strikingly, one mode of acquired resistance revealed therapeutic selection for a NEUROD1-high state with concomitant DLL3 downregulation. Other resistant tumors exhibited enrichment of regulatory and exhausted T-cell programs, highlighting tarlatamab's dual-targeting mechanism of action. Together, these results reveal that tarlatamab exerts selective pressure against ASCL1-driven lineages, facilitating resistance through loss of an antigen intrinsically linked to that state. These findings underscore the clinical relevance of TF-defined molecular subtypes in human SCLC. More broadly, they highlight the power of integrating longitudinal in vivo plasma transcriptional profiling from patient plasma with functional mouse modeling to uncover clinical and biological mechanisms of response and resistance to cell-surface-targeted therapies.