Neuroepithelial reprogramming and ERBB vulnerability in canine acanthomatous ameloblastoma
Neuroepithelial reprogramming and ERBB vulnerability in canine acanthomatous ameloblastoma
Stephanou, A.; Shui, B.; Mische, D.; Byron, M.; Katt, W. P.; Chan, M.; Grenier, J. K.; De Vlaminck, I.; Duhamel, G. E.; Gujral, T. S.; Sethupathy, P.; Peralta, S.
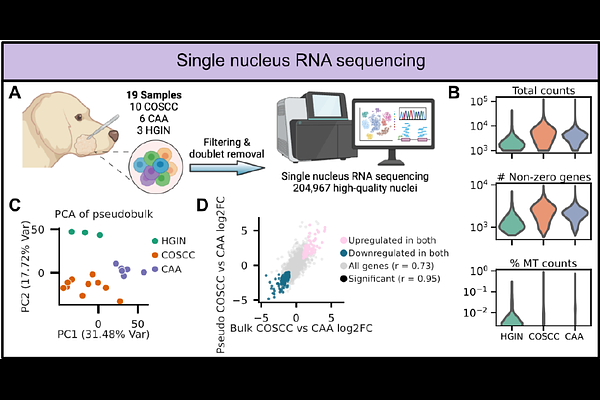
AbstractCanine acanthomatous ameloblastoma (CAA) is a locally invasive benign oral neoplasm that is difficult to distinguish from canine oral squamous cell carcinoma (COSCC) due to overlapping clinical, radiologic, and histologic features. Although both tumors exhibit MAPK pathway activation, their mutational landscapes are distinct. Furthermore, previous studies using bulk RNA sequencing (RNA-seq) have demonstrated pronounced differences in programs related to, among others, hypoxia, PI3K-AKT signaling, and cell proliferation. However, these bulk studies lacked the resolution to elucidate the cellular heterogeneity of CAA relative to COSCC. We therefore performed single-nucleus RNA-seq to define the cellular gene expression landscape of CAA, COSCC, and healthy gingiva. Across ~205,000 nuclei, we identified major epithelial, immune, endothelial, and mesenchymal populations, as well as two epithelial subtypes uniquely enriched in CAA. The CAA-specific keratinocytes exhibited a neuronal-like expression program defined by synaptic regulators, KRAS-associated signaling pathways, and markedly elevated expression of PEG3, ERBB4, GABRB1, MAGI2, and CASK. These findings were validated by bulk RNA-seq, qPCR, and immunohistochemistry, which demonstrated strong nuclear localization of PEG3 exclusively in CAA epithelium. A kinase inhibitor screen independently identified ERBB4 as a candidate therapeutic vulnerability, and pharmacologic inhibition with neratinib was effective. Together, these findings reveal a previously unrecognized neuroepithelial cell state that defines CAA, distinguishes it from COSCC, and reveals unique diagnostic and therapeutic signaling dependencies. Given the molecular and histopathologic parallels between CAA and human ameloblastoma, these data further position CAA as a naturally occurring comparative model for studying ameloblastoma biology and therapeutic vulnerabilities.