A guide for establishing patient-derived organoids from bile samples obtained during endoscopic procedures and performing gene expression knockdown
A guide for establishing patient-derived organoids from bile samples obtained during endoscopic procedures and performing gene expression knockdown
Rojo, C.; Vila, J. J.; Guembe, L.; Arrubla-Gamboa, A.; Jusue-Irurita, V.; Carrascosa-Gil, J.; Rullan, M.; Randez, J.; Fernandez-Barrena, M. G.; Huch, M.; Urman, J.; Avila, M. A.; Berasain, C.; Arechederra, M.
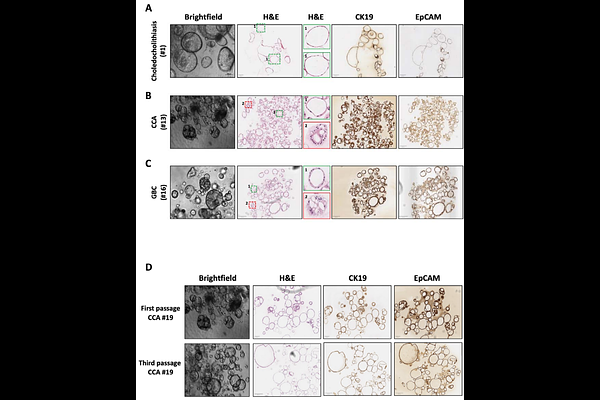
AbstractBile represents a clinically accessible biological fluid that can mitigates major limitations associated with tissue-based sampling for the generation of organoid models to study hepatobiliary disease, including biliary tract cancers where tissue availability is often limited. Importantly, bile can also enable the generation of non-malignant cholangiocyte organoids that are otherwise difficult to obtain. Here, we describe an operator-oriented, step-by-step protocol to generate organoids from fresh bile collected during endoscopic retrograde cholangiopancreatography (ERCP), together with two complementary workflows for siRNA delivery in 3D cultures. We detail critical control points that are often under-reported, yet considerably influence success and reproducibility. The protocol was optimized and applied in a real-world cohort of 21 patients undergoing ERCP, including benign biliary obstruction due to choledocholithiasis (n=5) and malignant strictures (n=16: cholangiocarcinoma n=13, gallbladder adenocarcinoma n=1, ampullary tumors n=2). Expandable organoids were established in 17/21 cases (81%), with establishment rates of 60% for choledocholithiasis and 85-100% across malignant entities. Anticipated results include organoid outgrowth within ~2-3 weeks and morphological heterogeneity in cultures derived from malignant strictures, where normal-like and tumor-like populations may initially coexist and can drift toward a cystic phenotype under routine expansion, motivating optional manual handpicking when tumor-enriched lines are required. As downstream readouts, we show feasibility of DNA-based profiling in selected paired bile-organoid samples (targeted sequencing and ULP-WGS copy-number analysis) and demonstrate proof-of-concept gene silencing via siRNA in both dissociated cells prior to re-embedding, and intact fully formed organoids while preserving 3D architecture. Collectively, this workflow provides a practical and reproducible framework to establish, expand, characterize and functionally perturb bile-derived organoids from routine clinical procedures, facilitating standardized implementation across laboratories.