Intranasal AAV Vaccination of SARS-CoV-2 Induce Strong and Sustained Neutralizing Antibodies in Mice
Intranasal AAV Vaccination of SARS-CoV-2 Induce Strong and Sustained Neutralizing Antibodies in Mice
Ji, Y.; Di Pasquale, G.; Zheng, C.; Afione, S.; Esperanza, T.; Yin, H.; Burbelo, P. D.; Chiorini, J. A.
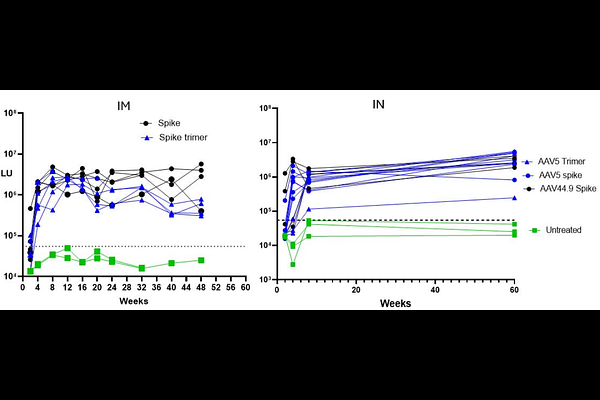
AbstractThe COVID-19 pandemic continues to pose significant health challenges, despite existing vaccines. This study evaluates the immunogenicity of recombinant adeno-associated viruses (AAV) expressing SARS-CoV-2 spike proteins, administered intramuscularly and intranasally in mice. Both delivery methods of AAV5-spike, AAV5-spike stabilized trimer as well as AAV44.9-spike elicited robust serum anti-spike antibodies within 8-12 weeks, with high levels of anti-spike antibodies sustained for over a year. Comparison of mouse serum antibodies 16 weeks post intramuscular or intranasal AAV5 administration demonstrated similar SARS-CoV-2 spike binding neutralizing activity in vitro. Analysis of changes in cellular immunity by ELISpot at 12 weeks post-AAV spike transduction revealed interferon-{gamma} induction in response to peptide challenge. Despite a decline in AAV vector DNA at the injection site, the persistence of anti-spike antibodies demonstrate that AAV-vectors can elicit lasting immune responses, highlighting nasal AAV administration as a potential strategy to block respiratory virus infections.