Metabolic reprogramming controlled by NF-YA alternative splicing creates therapeutic opportunities in colorectal cancer
Metabolic reprogramming controlled by NF-YA alternative splicing creates therapeutic opportunities in colorectal cancer
Belluti, S.; Mularoni, V.; Iseppato, N.; Campani, V.; Ronzio, M.; Righi, V.; Cuoghi, L.; Rinaldi, A.; Martinelli, T.; Cani, O.; Salsi, V.; Alessandrini, A.; Miserocchi, G.; Dolfini, D.; Zappavigna, V.; Imbriano, C.
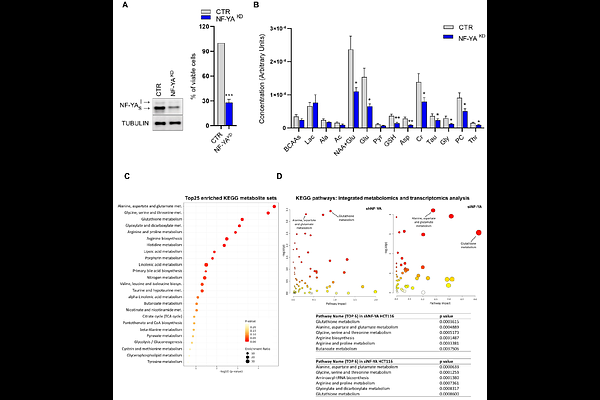
AbstractMetabolic reprogramming is a fundamental strategy that allows colorectal cancer (CRC) cells to endure microenvironmental constraints and sustain malignant progression. Here, we identify the transcription factor NF-Y as a master regulator of glutamine metabolism in CRC, with particular relevance to the aggressive CMS4 subtype. Loss of function experiments, integrated with metabolomic and transcriptomic analyses, reveal a critical role for NF-YA in regulating glutamine metabolism in CRC cells. Complementary gain of function studies pinpoint NF-YAl as the isoform specifically driving glutamine-centered rewiring. Mechanistically, NF-YAl directly binds the Glul promoter, inducing transcriptional upregulation of glutamine synthetase and increasing intracellular glutamine availability. This metabolic reprogramming enhances resistance to mechanical shear and oxidative stress under glutamine-limiting conditions, thereby promoting migratory and metastatic traits. Importantly, pharmacological inhibition of glutamine synthesis, but not uptake or downstream catabolism, selectively abrogates the survival and migratory advantage of NF-YAlhigh cells both in vitro and in vivo, highlighting a targetable vulnerability in aggressive CRC. Beyond CRC cell-autonomous advantage, NF-YAl-dependent glutamine biosynthesis reshapes the tumor microenvironment by promoting M2 macrophage polarization. Conditioned medium from NF-YAlhigh CRC cells is sufficient to induce human monocytes to adopt an M2-like phenotype. This effect is dependent on NF-YAlhigh tumor-derived glutamine, as inhibition of glutamine uptake by monocytes fully blocks their conversion to M2. In line with this, integrative analyses of patient-derived datasets underscore the predictive relevance of the NF-YAl-Glul-M2 axis in driving CRC aggressiveness. These findings define glutamine synthetase as a pivotal mediator of NF-YAl activity and a promising druggable metabolic Achilles' heel in NF-YAlhigh CRC tumors.