Simplifying principles that underlie the highly complex peptide motif of the promiscuous chicken class I molecule, BF2*21:01
Simplifying principles that underlie the highly complex peptide motif of the promiscuous chicken class I molecule, BF2*21:01
Harrison, M.; Chappell, P. E.; Halabi, S.; Danysz, M.; Mararo, E. M.; Magiera, L.; Hermann, C.; Deery, M. J.; Lilley, K. S.; Wallny, H.-J.; Avila, D. W.; Mwangi, W.; Nair, V.; Lea, S. M.; Ternette, N.; Kaufman, J.
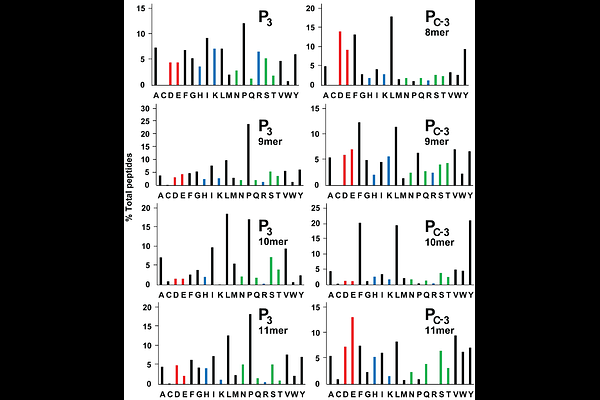
AbstractIn chickens and humans, classical class I molecules of the major histocompatibility complex (MHC) can have a hierarchy of correlated properties, including cell surface expression and peptide repertoire. Chicken BF2 alleles that are less well-expressed on the cell surface and bind a very wide range of peptides are expressed by MHC haplotypes that confer protection from a variety of economically-important infectious diseases, while certain human HLA-B alleles that are well-expressed and bind a narrow range of peptides lead to slow progression from HIV infection to AIDS. Understanding the impact of these promiscuous generalists and fastidious specialists is of considerable interest. The promiscuous BF2 molecule from the chicken B21 haplotype, BF2*21:01, binds a wide range of peptides by remodelling the peptide-binding site, allowing co-variation of the anchor residues at peptide positions P2 and Pc-2, and binding of an anchor residue at Pc. By using in vitro refolding assays with peptides and peptide libraries, determining thermostability and crystal structures, and analysing a chicken B21 cell line by immunopeptidomics, we found that BF2*21:01 will accommodate many possible combinations at P2 and Pc-2, as well as several hydrophobic amino acids at Pc. However, marked preferences for particular peptide lengths, particular amino acids at the three anchor residues, combinations of amino acids at P2 and Pc-2, and amino acids at P3 and Pc-3 affecting stability lead to high frequencies of major peptides while still allowing the possibility of presenting a wide peptide repertoire. These simplifying principles may eventually allow predictions of pathogen peptides with stable binding for this iconic promiscuous class I molecule, as well as providing the data for more sophisticated peptide prediction methods.