Patient-derived organoids from malignant pleural effusion to explore for alternative therapies in thoracic tumors
Patient-derived organoids from malignant pleural effusion to explore for alternative therapies in thoracic tumors
Ferreiro-Miguens, R.; Diez-Grandio, I.; Soto-Feijoo, R.; Ferreiro, L.; Garcia, J.; Otero-Alen, M.; Abdulkader, I.; Bernandez, B.; Dominguez, E.; Abal, M.; Leon-Mateos, L.
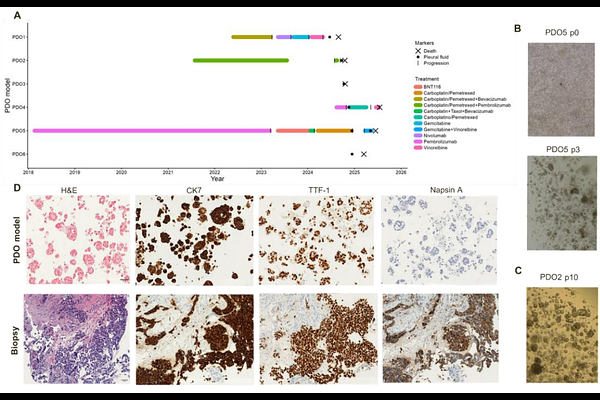
AbstractThoracic malignancies, including lung adenocarcinoma (ADC) and malignant pleural mesothelioma (MPM), remain associated with poor prognosis and limited durable therapeutic responses in advanced stages. Although targeted therapies and immunotherapy have improved outcomes in selected patients, systemic chemotherapy continues to play a central role in routine clinical practice. However, treatment response is highly heterogeneous, and reliable predictive biomarkers of chemotherapy sensitivity are lacking. Both ADC and MPM frequently involve the pleural cavity and are commonly associated with malignant pleural effusion (MPE), which contributes to symptoms such as dyspnea and chest pain and requires therapeutic drainage. Importantly, MPE represents a clinically accessible source of viable tumor cells obtained through minimally invasive procedures. In this study, we established patient-derived organoids (PDOs) from malignant pleural effusion samples obtained from five patients with advanced lung adenocarcinoma and, as an exploratory extension, from one patient with malignant pleural mesothelioma. Organoids were characterized by immunohistochemistry and subjected to systematic chemotherapy drug screening. Inter-model variability in treatment response was assessed, and selected drug sensitivities were further validated through dose-response assays. Pleural effusion-derived organoids successfully recapitulated tumor-specific phenotypic features and revealed marked heterogeneity in chemotherapy sensitivity across models. Secondary validation confirmed the reproducibility of selected responses. Our findings support the feasibility of generating functional organoid models from malignant pleural effusions and highlight their potential as translational platforms for individualized chemotherapy profiling in advanced thoracic malignancies.