A tumor metabolism-angiogenesis-immune axis governs immunotherapy responses
A tumor metabolism-angiogenesis-immune axis governs immunotherapy responses
Serganova, I.; Colombo, G.; Ballesio, F.; Kang, J. H.; Karakousi, T.; Esposito, T. V. F.; Ackerstaff, E.; Santella, A.; Blasberg, R.; Pillarsetty, N. V. K.; Schreier, A.; Andreopoulou, E.; Demaria, S.; Lund, A. W.; Gherardini, P. F.; Zappasodi, R.
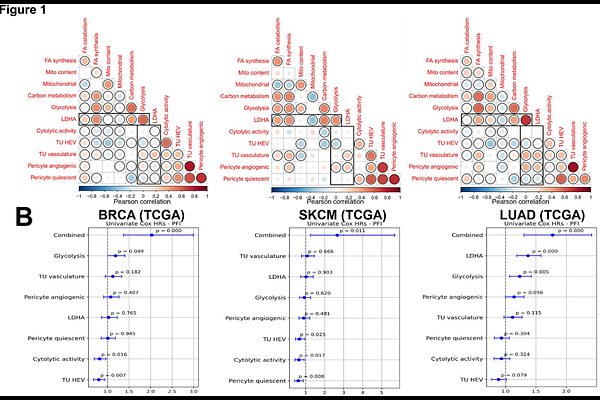
AbstractDespite the clinical success of immunotherapy, long-lasting benefit remains restricted to a subset of patients. Tumor metabolic adaptation is emerging as a key factor limiting immunotherapy efficacy. We previously found that glycolysis-low tumor variants, compared to the parental glycolytic tumors, better respond to neoadjuvant CTLA-4 immune checkpoint blockade (ICB) therapy. Here, we investigated new rational modalities to restore immune sensitivity of glycolytic tumors by studying how lowering the tumor-cell glycolytic capacity reshapes the tumor microenvironment (TME) to favor long-lasting systemic anti-tumor responses upon immunotherapy. We found that lowering glycolysis in cancer cells through LDHA-knock down (KD) results in TME displaying normalized vasculature, reduced angiogenic markers, increased high endothelial venules (HEVs), and enhanced recirculation of CD8+ T cells both in and out of the tumor. By leveraging public transcriptomic data sets from human solid cancers, we confirmed that glycolysis positively correlates with neo-angiogenesis and inversely correlates with features of vascular normalization and immune cytolytic activity. Moreover, a tumor signature that incorporates glycolysis- and angiogenesis-related genes as positive features, and normal vasculature, HEV, and immune cytolytic activity genes as negative features predicted poor outcomes better than the individual features across most human solid tumor types in the TCGA. To determine the therapeutic implication of these interrelated processes, we asked if targeting the vasculature would restore immunotherapy responses in glycolytic tumors. We found that combining low-dose anti-VEGFR2 with CTLA-4 blockade induces tumor regressions and protection from metastases in glycolytic tumors. These therapeutic effects were associated with vasculature normalization and increased abundance of HEVs and concentrations of lymphangiogenic factors in the TME of glycolytic tumors. Moreover, anti-VEGFR2 with anti-CTLA-4 restored recirculation of anti-tumor CD8+ T cells in and out of the TME in glycolytic tumors, with specific increases in intratumoral recruitment and activation of cytolytic CD62LCD44CD8 T cells expressing VEGFR2 and low levels of CTLA-4, suggesting potential novel direct synergistic effects of anti-VEGFR2 and anti-CTLA-4 on CD8+ T cells. Conversely, this combination opposed the beneficial immune and vascular TME features of LDHA-KD tumors, indicating tumor-metabolic-dependent effects. Accordingly, we found that standard combined regimens of anti-VEGF and ICB therapy improve survival with respect to ICB alone in patients with glycolysis-high but not glycolysis-low tumors. Together, these findings indicate that tumor cell glycolysis primes the TME for aberrant vascular architecture and T-cell exclusion, and that modulating the tumor vasculature can unravel these mechanisms restoring immune responsiveness. This suggests that tailoring anti-angiogenic and immunotherapy combinations to the tumor glycolytic state and associated vasculature profiles may restore immune surveillance and overcome therapy resistance.