Meditope-Enabled Chimeric Antigen Receptors Facilitate Plug-and-Play Control of T Cells
Meditope-Enabled Chimeric Antigen Receptors Facilitate Plug-and-Play Control of T Cells
Kuo, C.-F.; Tong, Z.; Kuo, Y.-C.; Kuo, M.; King, J.; Ly, K.; Parcutela, B.; Stern, L. A.; Wang, Z.; Aguilar, B.; Starr, R.; Chang, W.-C.; Ostberg, J. R.; Rossi, D.; Clark, M. C.; Alizadeh, D.; Forman, S. J.; Williams, J. C.; Brown, C. E.
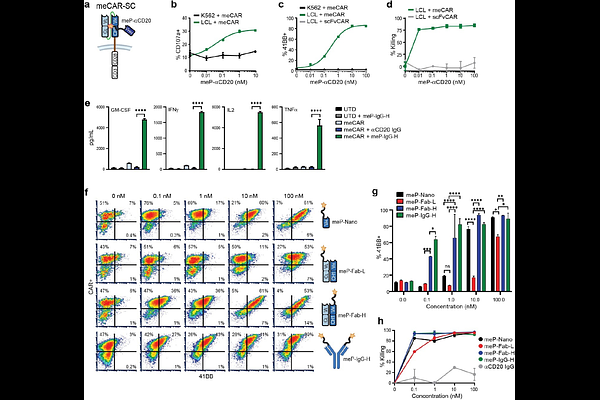
AbstractChimeric antigen receptor (CAR) T cells have transformed cancer treatment, yet challenges for achieving broader clinical success remain, including overcoming tumor antigen heterogeneity and limited T cell fitness. To address these challenges and enhance CAR T cell functionality, we leveraged meditope technology, a lock-and-key platform where Fab regions of antibodies are modified to bind a small cyclic peptide termed meditope (meP). We developed a panel of meditope-enabled Fab-based CARs (meCARs), which show selective binding to the meP and comparable activity to traditional single-chain variable fragment (scFv)-based CARs. Focusing on HER2-targeted meCARs for evaluating platform utility, we exploited the modularity of the meditope platform to detect meCAR T cells using meP-fused fluorescent agents, promote meCAR T cell expansion via meP-fused IL-15 cytokine, and broaden tumor antigen targeting through meP-fused antibodies to address tumor heterogeneity. These findings establish the meditope technology as a versatile strategy to augment CAR T cell functionality and overcome key limitations of current CAR-based therapies.