Mechanical memory of confinement pressure governs expansion size in epithelial monolayers
Mechanical memory of confinement pressure governs expansion size in epithelial monolayers
Engstrom, L.; Schnyder, S. K.; Kumra Ahnlide, J.; Grudtsyna, V.; Gloerich, M.; Nordenfelt, P.; Doostmohammadi, A.; Swaminathan, V. S.
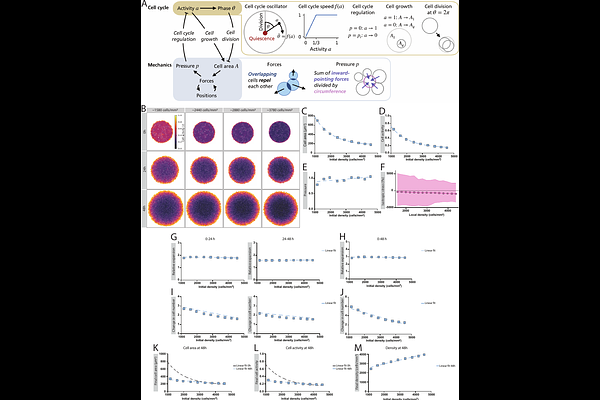
AbstractEpithelial tissues undergo rapid expansion during development, repair, and morphogenesis, yet how tissue-scale growth is coordinated to re-establish homeostasis remains unclear. Here, we show that large epithelial monolayers confined at a wide range of initial densities and mechanochemical states robustly converge to the same final size and density upon release, despite differences in initial cell size, YAP activity, and cell number dynamics. To investigate the underlying mechanism, we combined quantitative experiments with a mechanochemical agent-based model in which mechanical pressure arising from confinement acts as a tissue-scale signal that modulates intracellular cell-cycle activity over time. Using this framework, we show that transient mechanical relaxation during confinement selectively elevates cell-cycle activity in higher-density tissues at the time of release, accelerating early expansion without disrupting final homeostatic outcomes. Together, these results reveal how epithelial tissues coordinate collective growth and robustly restore homeostasis during expansion.