Energy-precision trade-off in mitotic oscillators revealed by ATP modulation in artificial cells
Energy-precision trade-off in mitotic oscillators revealed by ATP modulation in artificial cells
Wang, S.; Yourston, L.; Maryu, G.; Kim, Y.; Walker, D.; Kadiyala, U.; Yang, Q.
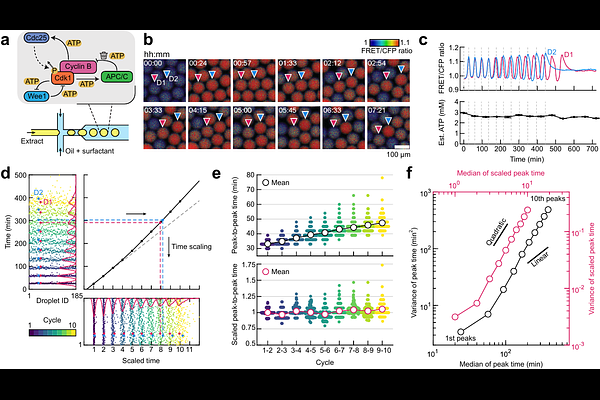
AbstractThe temporal precision of biochemical oscillators is fundamentally constrained by the energy dissipated to suppress molecular fluctuations, a widely predicted trade-off governing information processing across biology and physics, from molecular motors to kinetic proofreading to computing. Yet, experimental validation in complex biological oscillators remains elusive due to challenges of systematically modulating energy while quantifying stochastic dynamics across large ensembles. Here, we establish a high-throughput droplet-microfluidics platform to reconstitute mitotic oscillations from Xenopus laevis egg extracts within thousands of sub-nanoliter compartments. By precisely tuning ATP across a broad free-energy landscape and developing an analytical framework that decouples intrinsic phase diffusion from quenched period heterogeneity, we uncover a hidden trade-off linking metabolic budget, oscillation speed, and precision. While speed peaks non-monotonically near physiological ATP levels and declines toward both high and low bifurcation limits, precision increases monotonically with energy. These findings provide direct experimental evidence that mitotic timing is actively shaped by energy budgets. Intriguingly, embryonic cell cycles are not optimized for maximum fidelity, but for a metabolic compromise maintaining just enough coherence for synchronous yet rapid divisions, placing the endogenous ATP budget near an energetic optimum balancing speed and accuracy. Our integrated artificial-cell and analytical strategy provides a generalizable framework for mapping thermodynamic limits in non-equilibrium biological dynamics.