An Investigation of the Conformational Dynamics of ABC Exporter PCAT1 using Microsecond-Level MD Simulations
An Investigation of the Conformational Dynamics of ABC Exporter PCAT1 using Microsecond-Level MD Simulations
Brownd, M.; Khodadadi, E.; Moradi, M.
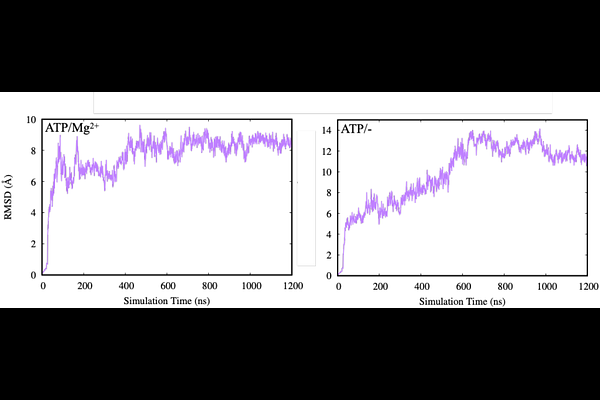
AbstractPeptidase-containing ATP-binding cassette transporters (PCATs) couple ATP hydrolysis with proteolytic processing and export of cargo peptides across cellular membranes. Despite their importance in bacterial secretion systems, the molecular determinants governing nucleotide binding and stabilization in PCAT transporters remain incompletely understood. In particular, recent experimental observations suggest that PCAT1 may display altered nucleotide preferences compared with canonical ABC transporters. Here, we employed microsecond-scale all-atom molecular dynamics simulations combined with free energy perturbation (FEP) calculations to characterize nucleotide binding, protein stability, and conformational dynamics of PCAT1 across multiple biochemical conditions. Simulations were performed for inward-facing (IF) and outward-facing (OF) conformations in the presence or absence of Mg2+ and substrate peptides. Structural analyses reveal that substrate and Mg2+ jointly stabilize the IF conformation, reducing global structural fluctuations and enhancing nucleotide retention in the binding pockets. In contrast, systems lacking Mg2+ exhibit increased nucleotide mobility and partial dissociation events. Thermodynamic analysis using FEP calculations further demonstrates that ATP binding is strongly stabilized in the IF state, particularly in the presence of Mg2+, whereas nucleotide stability is reduced when Mg2+ coordination is absent. To identify the molecular origins of nucleotide stabilization, we introduce a residue-level free energy decomposition approach that quantifies the contribution of individual residues to nucleotide binding energetics. This analysis reveals that the Walker A residue Lys525 provides the dominant stabilizing interaction with ATP, while neighboring residues within the Walker A motif contribute additional stabilization. In contrast, acidic residues of the Walker B motif primarily participate in catalytic organization rather than direct nucleotide stabilization. Together, these results provide a comprehensive molecular description of nucleotide stabilization and conformational regulation in PCAT1. The combined structural and energetic analyses support a model in which Mg2+ coordination and substrate binding cooperatively stabilize the inward-facing state and organize the nucleotide-binding site for productive ATP hydrolysis. More broadly, this work demonstrates how residue-level free energy analysis can reveal the energetic architecture of nucleotide recognition in ABC transporters.