A framework for testing structural hypotheses of protein dynamics against experimental HDX-MS data
A framework for testing structural hypotheses of protein dynamics against experimental HDX-MS data
Siddiqui, A. I. H.; Skyner, R.; Musgaard, M.; Krishnamurthy, S.; Deane, C.; Crook, O.
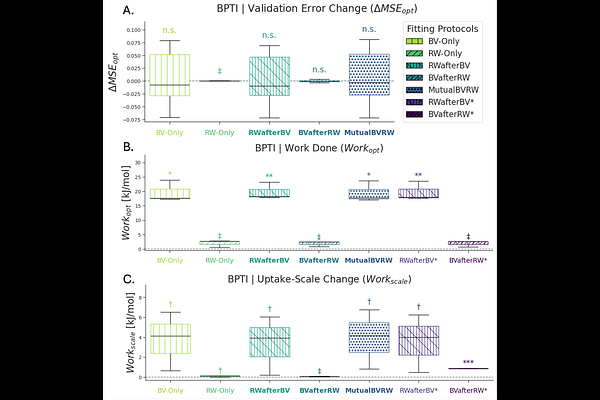
AbstractProtein dynamics determine biological function, yet extracting structural ensembles from Hydrogen-Deuterium Exchange Mass Spectrometry (HDX-MS) remains a challenging inverse problem. Current ensemble-fitting approaches often achieve good agreement with uptake curves but lack rigorous validation and uncertainty quantification, limiting structural confidence. We propose ValDX, a validation framework for quantitative integration of HDX-MS data with structural ensembles. ValDX combines overlap-aware data splitting, replicate-based uncertainty estimation, and uptake-independent "Work Done" metrics that quantify how much an ensemble must be modified to match experiment. Across 22 ensembles spanning six proteins (58-474 residues), we show that conventional error metrics fail to distinguish structurally representative ensembles from incorrect ones, whereas Work Done metrics robustly discriminate global and local conformational quality. We further demonstrate that clustering yields compact, interpretable ensembles with minimal loss of accuracy, and that staged optimisation enables reliable fitting of both ensemble weights and forward-model parameters without requiring a reference structure. Together, this framework establishes HDX-MS ensemble integration as a quantitative structural hypothesis-testing problem, enabling inference of protein dynamics from HDX-MS data.