Discovery and in vivo characterization of novel TOG domain-containing proteins using C. elegans
Discovery and in vivo characterization of novel TOG domain-containing proteins using C. elegans
Wethekam, L. C.; Mittal, T.; Maddox, A. S.
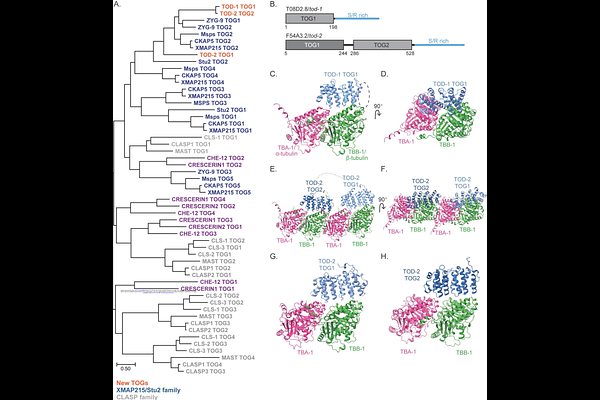
AbstractThe proper assembly, architecture, and maintenance of microtubule, actin and other cytoskeletal networks require regulation by various polymer binding proteins. Microtubules, rely on both the tubulin building blocks, but also many tubulin- and microtubule binding proteins. TOG domain-containing proteins comprise one family of tubulin-binding proteins that regulate microtubule dynamics. Here we identify two previously uncharacterized TOG domain-containing proteins (TOD-1 and TOD-2) in the nematode, C. elegans. These proteins are unique in that they are members of the XMAP215 family but contain reduced numbers of TOG domains and, in one case, a divergent TOG domain. TOD-1 and TOD-2 are expressed in and contribute to the normal function of sperm. The single TOG domain of TOD-1 and both TOG domains of TOD-2 are predicted to bind free tubulin dimers and not microtubule lattice. Deletion of either tod gene resulted in an increased laying of unfertilized oocytes. Inspection of mutant hermaphrodites revealed a premature onset of sperm migration failure. Together, these findings suggest that C. elegans requires regulation of tubulin dimers and/or microtubules for sperm localization and function. The amoeboid movement of C. elegans sperm has been considered microtubule-independent, our results open a new avenue of research into their unique motility.