Dosage compensation defects due to roX RNA deletion are rescued by recalibration of X/autosome stoichiometry
Dosage compensation defects due to roX RNA deletion are rescued by recalibration of X/autosome stoichiometry
Gkountromichos, F.; Yankson, G.; Jayakrishnan, M.; Campos Sparr, A.; Müller, M.; Heun, P.; Becker, P. B.
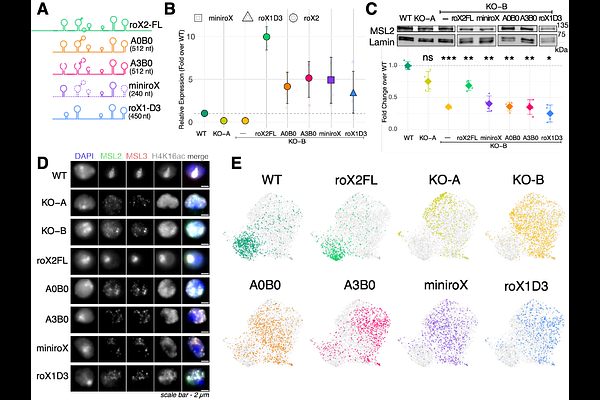
AbstractMetazoa evolved regulatory networks to balance the expression of their sex chromosomes. In Drosophila, males have a single gene-rich X chromosome, whereas females have two. Balanced X/autosome expression is essential for viability, and in male flies is achieved by activation of genes on the X through the male-specific-lethal (MSL) dosage compensation complex (DCC). This ribonucleoprotein assembly contains long, non-coding roX RNAs. To dissect the functional requirements of roX in a cell-based system, we deleted the roX2 gene in male S2 cells and selected two independent lines lacking detectable roX RNA. In the absence of roX, the remaining MSL protein complex was unable to associate with known or newly identified binding sites and thus failed to activate transcription. Surprisingly, the X/autosome expression ratio appeared nevertheless compensated. Cytogenetic and genomic analyses revealed that both roX-deficient cell populations had acquired additional X chromosomes. Apparently, chromosome gains due to mis-segregation made up for the loss of DCC-mediated dosage compensation. Interestingly, ectopic expression of full-length roX2, but not of shortened derivatives, fully restored DCC binding and normalized the karyotype. These findings illustrate that X chromosome dosage compensation is critical for viability even in cultured cells and provide a striking example of rapid evolution under stringent selection.