Nuclear βactin dependent chromatin accessibility governs stem cell pluripotency and extracellular matrix gene programs to maintain cellular biomechanics for cell lineage decisions
Nuclear βactin dependent chromatin accessibility governs stem cell pluripotency and extracellular matrix gene programs to maintain cellular biomechanics for cell lineage decisions
Campbell, C.; Hosny El Said, N.; Ghassan Al Nuairi, A.; Loganathan, P.; Breunig, C.; Deliorman, M.; Desbordes, S. C.; Lohse, M. J.; Magzoub, M.; Qasaimeh, M. A.; Percipalle, P.
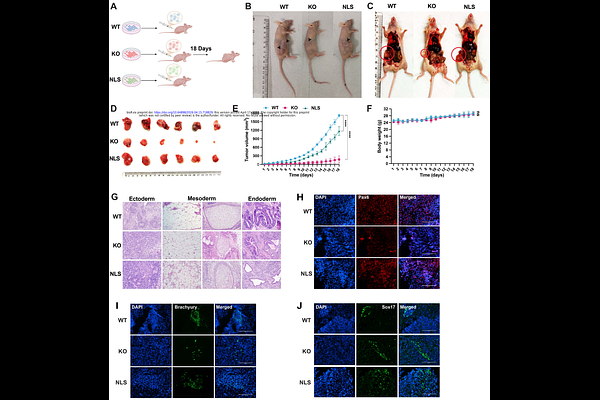
AbstractPluripotency and lineage commitment in embryonic stem cells emerge from transcriptional programs shaped by higher-order chromatin architecture and the integration of extracellular matrix (ECM) cues, yet the molecular basis of this coordination remains unresolved. Here we identify nuclear {beta}-actin as a central regulator coupling chromatin organization to ECM-dependent control of cell fate in mouse embryonic stem cells. CRISPR/Cas9-mediated ablation of {beta}-actin leads to loss of core pluripotency factors, including Oct4 and Sox2, and drives widespread transcriptional reprogramming, whereas nuclear-targeted re-expression restores these defects. Genome-wide chromatin accessibility profiling reveals a pronounced reduction at regulatory elements of pluripotency genes, consistent with impaired chromatin remodeling. In parallel, ECM gene programs are aberrantly activated, resulting in altered matrix composition, increased stiffness heterogeneity and disrupted cellular biomechanics. Functionally, these changes compromise self-renewal, bias lineage specification and impair differentiation capacity, notably blocking neuronal differentiation while promoting mesodermal-like fates. In vivo, {beta}-actin depletion markedly restricts teratoma growth and disrupts tri-lineage potential. Reintroduction of nuclear {beta}-actin restores chromatin accessibility, transcriptional programs, ECM properties and differentiation competence. Together, our findings position nuclear {beta}-actin as a key integrator of genome architecture, mechanotransduction and transcription, thereby linking chromatin state to ECM-dependent regulation of stem cell identity.