MauE from Calditrichota and Thermodesulfobacteriota reveal a new pathway for disulfide bond formation in bacteria
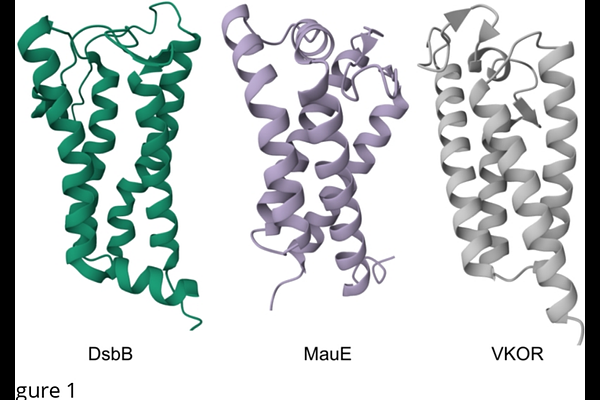
MauE from Calditrichota and Thermodesulfobacteriota reveal a new pathway for disulfide bond formation in bacteria
Gonzalez, C.; Moilanen, A.; Korhonen, K.; Thu, N. P. A.; Hiltunen, J.; Saaranen, M.; Ruddock, L. W.
AbstractDisulfide bond formation is crucial to the structure and function of many proteins. It is known that there is diversity in the pathways for disulfide bond formation in bacteria and that there are gaps in our knowledge of these pathways. Using a combination of experimental and bioinformatic approaches we show that some of these gaps can be filled by a newly discovered oxidative folding pathway centered on methylamine utilization protein E (MauE). MauE has previously been associated with the methylamine utilization (MAU) gene cluster, which is involved in methylamine metabolism, in particular it is associated with the maturation of the small subunit of methylamine dehydrogenase. Here we show MauE from Caldithrixabyssi and Desulfatibacillum alphaticivorans functionally replace disulfide bond formation protein B (DsbB) in E. coli using two independent disulfide bond dependent assays. Furthermore, MauE is found in 14 species from 2 bacterial phyla that lack known pathways for structural disulfide bond formation, but which have proteins with structural disulfide bonds in the protein data bank. The active site for MauE was determined to be a conserved CXC motif. Using molecular docking predictions, we demonstrate that MauE is likely to interact with ubiquinone, similarly to the well characterized bacterial DsbB. We also constructed a dataset across thirty-five different phyla to demonstrate that MauE is potentially the second most common disulfide bond formation protein in bacterial disulfide bond formation pathways after DsbB. In addition, the distribution of MauE largely differs from the distribution of other MAU gene cluster markers affirming its role as a newly discovered generalist disulfide bond formation protein rather than being a specialized maturation factor for methylamine dehydrogenase. We also reveal further gaps in disulfide bond pathways, as well as species which may contain redundancies in their disulfide bond pathways.