Ritonavir-Induced Cellular Stress Alters Viral HBs Glycoprotein Biogenesis and Production of Infectious Hepatitis D Virions
Ritonavir-Induced Cellular Stress Alters Viral HBs Glycoprotein Biogenesis and Production of Infectious Hepatitis D Virions
El orch, W.; Vidalain, P.-O.; Jacquemin, C.; Durantel, D.; Pastor, F.; Barnault, R.; Charriaud, F.; wuilbaut, a.; Nabaile, C.; Kadokura, H.; Gaetani, M.; BEGUE, M.; RICHARD, E.; Sureau, C.; Verrier, B.; Ramiere, C.; JANIN, Y.; Lotteau, V.; deleuze, c.; Michel, M.
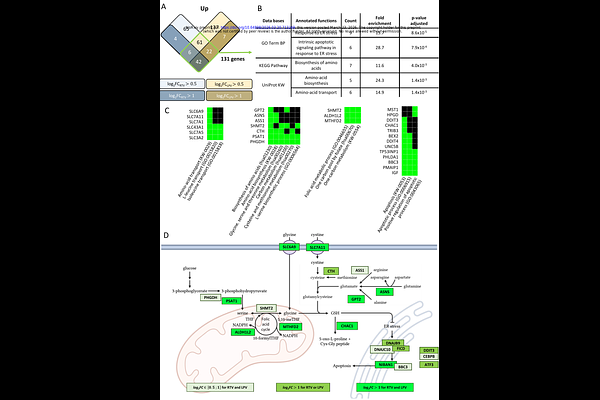
AbstractChronic co-infections by HBV and its satellite virus HDV are associated with a high risk of progression to cirrhosis and liver cancer, and therapeutic options for achieving a cure are still unsatisfactory. HBs is the main surface glycoprotein of both viruses, and is also massively secreted by infected hepatocytes in the form of empty subviral particles which suppress the host immune responses. This makes HBs an attractive target to develop therapeutic strategies. Here, we took advantage of the known interaction between the Large form HDV antigen (HDAg-L) and the small form of HBs (S-HBs) to develop a non-infectious, minimalistic reporter assay for the assembly and secretion of HDV particles. By screening the existing pharmacopeia for drugs that could interfere with S-HBs and HDAg-L co-secretion, we found that ritonavir and other Cytochrome P450 inhibitors affect the biogenesis of HBs and impair the production of infectious HDV virions. Mechanistically, we established that these drugs induce oxidative stress which dysregulates disulfide bond formation in the endoplasmic reticulum. As a consequence, the production of HBs, which depends on a dense network of disulfide bonds, is markedly affected as evidenced by an abnormal glycosylation profile, altered antigenic properties, and a poor expression of the largest form of HBs (L-HBs) which is essential to virus entry into target cells. This is associated with induction of the unfolded protein response, with the upregulation of CHOP/DDIT3 and key enzymes involved in the synthesis of the reducing metabolite glutathione (PHGDH, SHMT2, MTHFD2). Overall, our results indicate that alterations in redox homeostasis significantly impact HBs biogenesis, and reveal a druggable pathway that could be exploited to eliminate HDV in chronically infected patients.