NLRP3 activators disrupt the endocytic AP2 complex and plasma membrane signaling
NLRP3 activators disrupt the endocytic AP2 complex and plasma membrane signaling
Ebner, S.; Phulphagar, K. M.; Alvarez, Y.; Juergenliemke, L.; Frechen, F.; Stoetzel, I.; Lovotti, M.; Mangan, M. S. J.; Akbal, A.; Schneberger, N.; Frauenstein, A.; Klein, T.; Swietlik, J. J.; Gansen, B. O.; Fink, D.; Lindner, A. U.; Nguyen, L.; Becher, I.; Walter, J.; Rollheiser, J.; Kudaliyanage, A.; Graetz, L.; Gerken, O. J.; Itzhak, D.; Borner, G. H. H.; Tanzer, M. C.; Duthie, F.; Stahl, R.; Kallabis, S.; Will, D.; Savitski, M. M.; Schroeder, A.; Jantsch, J.; Hagelueken, G.; Legler, D. F.; Mann, M.; Latz, E.; Kiermaier, E.; Wachten, D.; Kostenis, E.; Meissner, F.
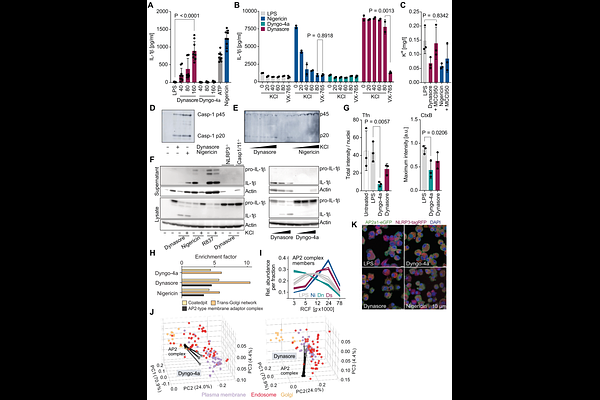
AbstractOrganellar perturbations are linked to NLRP3 inflammasome activation, however, it remains unclear whether unrelated agonists converge on a common upstream pathway. Here, we traced intracellular organelle and protein movements by differential ultracentrifugation combined with mass spectrometry-based proteomics. We show that NLRP3 activators uniformly disrupt the endocytic Adaptor Protein 2 (AP2) complex, whereas other subcellular rearrangements are stimulus-specific. We discovered Dynasore as a K+ efflux-independent NLRP3 activator that engages this signaling node irrespective of endocytosis inhibition. Pharmacological and genetic perturbation of AP2 renders cells unresponsive to extracellular cues, blunting GPCR signaling, cAMP production, and chemotaxis, thereby enforcing a 'frozen' signaling state that propagates NLRP3 inflammasome activation. Collectively, our study reveals a common surveillance checkpoint linking impaired plasma membrane signaling to the execution of inflammation and cell death.