A full-length single nuclei transcriptomic atlas of human skeletal muscle insulin resistance
A full-length single nuclei transcriptomic atlas of human skeletal muscle insulin resistance
Whytock, K. L.; Divoux, A.; Vazquez, J.; Hopf, M.; Viggars, M. R.; Gutierrez-Monreal, M. A.; Ruggiero, C. H.; Jimenez-Rondan, F. R.; Morena, F.; Krassovskaia, P.; Broskey, N. T.; Sun, Y.; Walsh, M. J.; Cousins, R. J.; Houmard, J. A.; Sparks, L. M.; Goodpaster, B. H.
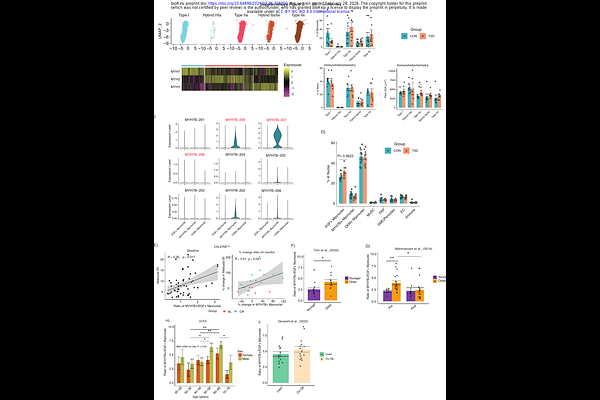
AbstractSkeletal muscle (SkM) insulin resistance is a central defect in T2D, yet cell specific molecular determinants remain incompletely understood. Here, we integrate full-length single-nucleus transcriptomics with gold-standard stable isotope-labeled hyperinsulinemic-euglycemic clamps to generate a nucleus-resolved transcriptomic atlas of SkM insulin resistance. We identify previously unrecognized myonuclear populations whose proportions associate with insulin sensitivity across independent cohorts, revealing MYH7B+ myonuclei are metabolically favorable over EGF+ myonuclei. Modeling transcriptional variation against tracer-derived glucose disposal uncovers highly nucleus-specific molecular programs that are obscured when using surrogate fasting indices. Mechanistically, we identify zinc transporter ZIP14 as a positive regulator of insulin-stimulated glucose uptake and implicate EGF signaling in impaired branched-chain amino acid catabolism and inflammatory cross-talk within the SkM niche. Together, these findings redefine SkM insulin resistance as a multicellular, nucleus-resolved process and highlight new cell type specific targets for metabolic intervention.