Selective targeting of a histone-like silencer Sfx to the R6K conjugal transfer operon
Selective targeting of a histone-like silencer Sfx to the R6K conjugal transfer operon
Wang, B.; Gupta, R.; Blaine, N.; Khitiri, B.; Jordan, C.; Molotievskiy, N.; Dunlap, D.; Finzi, L.; Artsimovitch, I.
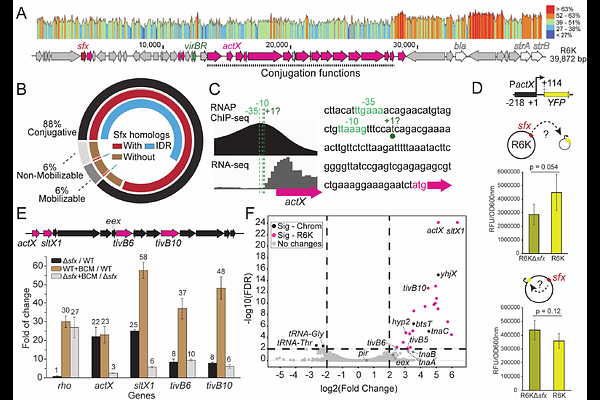
AbstractConjugative plasmids drive bacterial evolution and antibiotic resistance spread, yet their gene expression must be silenced to protect the host. A histone-like protein H-NS represses many mobile and sedentary xenogenes but fails to silence the conjugal transfer vir operon of R6K, a prototype IncX plasmid. Instead, R6K encodes its own H-NS homolog, Sfx, to repress the vir operon. Here, we show that, unlike other plasmid silencers that target promoters, Sfx cooperates with Rho factor to arrest transcription elongation. ChIP-seq reveals that despite sharing similar DNA motifs and a preference for negative supercoiling, Sfx and H-NS occupy distinct niches: Sfx binds weakly to the chromosome but is enriched on the R6K vir operon, from which H-NS is excluded. We hypothesize that this selective targeting is mediated by Sfx-vir interactions and phase separation. We show that Sfx binding to vir DNA critically depends on DNA topology but not on the target location. Our results suggest that Sfx phase separates with R6K to ensure its preferential recruitment to the plasmid DNA and forms stable nucleoprotein filaments that are impermeable to competitors. These findings reveal how histone-like proteins can partition the genome into distinct regulatory niches, a strategy likely mirrored across all life.