Δ⁹-Tetrahydrocannabinol-induced enhancement of reward responsivity via mesocorticolimbic modulation in squirrel monkeys
Δ⁹-Tetrahydrocannabinol-induced enhancement of reward responsivity via mesocorticolimbic modulation in squirrel monkeys
Hur, K.-H.; Nickerson, L. D.; Bergman, J.; Stover, J.; Kohut, S. J.
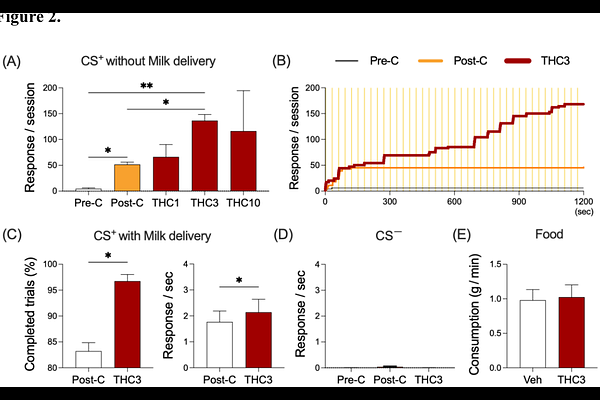
AbstractDelta-9-tetrahydrocannabinol (THC)-containing products are widely used recreationally, partly due to THC's ability to enhance the appetitive (i.e., rewarding) properties of diverse stimuli. However, the neural mechanisms through which THC modulates reward-related processing remain poorly understood. Here, we used a Pavlovian paradigm in adult squirrel monkeys (3males, 1female) to associate a visual conditioned stimulus (CS+) with appetitive food delivery. The modulatory effects of acute THC (1-10g/kg, i.m.) on behavioral and brain responses to CS+ were evaluated. Event-related functional MRI (fMRI) was employed to characterize the neural correlates of conditioned responding to the CS+, both in the absence and presence of THC treatment, with pre-conditioning scans serving as control. Behaviorally, THC (3g/kg) selectively enhanced conditioned responding to the CS+ without altering responses to the control stimulus (CS-) or increasing baseline consummatory responding, underscoring the specificity of THC's action on reward-associated processes. Consistently, fMRI analyses revealed that THC amplified CS+-evoked activation within mesocorticolimbic regions, including the anterior cingulate cortex (ACC), striatum, hippocampus, and substantia nigra-ventral tegmental area (SN-VTA), without affecting activity in visual and motor cortices. This finding underscores the selectivity of THC's neuromodulatory effects on reward-related circuitry. Independent of CS exposure, resting-state functional connectivity analyses indicate that THC enhanced mesocorticolimbic network integration, as evident in strengthened SN-VTA-centered connectivity with the ACC, striatum, and hippocampus. Collectively, these findings demonstrate that THC enhances the responses to appetitive stimuli, through selective modulation of mesocorticolimbic circuitry, highlighting the SN-VTA as a pivotal hub for cannabinoid-mediated regulation of incentive salience and motivational drive toward reward-associated stimuli.