A class of metallohydrolases expands bile salt hydrolase activity in the gut
A class of metallohydrolases expands bile salt hydrolase activity in the gut
Malarney, K. P.; Scott, S. A.; Chang, P. V.
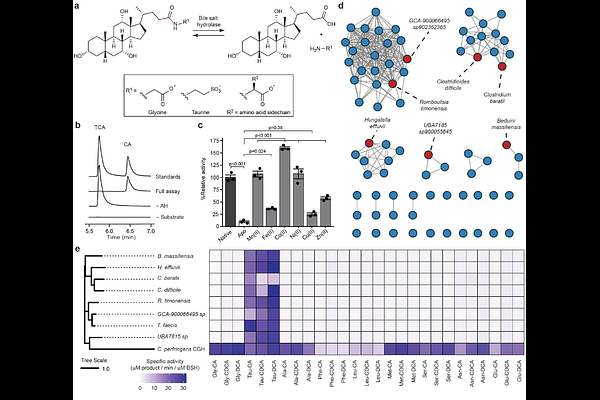
AbstractBile acids are steroidal metabolites produced by host and microbial metabolism that shape gut microbiome ecology and influence host physiology1,2. Bile acid structural diversification requires gut microbial bile salt hydrolase (BSH) activities, which cleave the amide bond of liver-derived bile acid amidates (BAAs)3. Beyond this gatekeeping metabolic function, BSHs expand the bile acid pool via their amine N-acyltransferase activity to produce numerous microbially derived BAAs that signal via host receptors and are further metabolized4,5. To date, all BSH activity has been attributed to a family of N-terminal nucleophile (Ntn) cysteine hydrolases. However, numerous gut anaerobic bacteria possess BSH activity but do not encode bsh genes from the Ntn family. Here, we describe a previously unknown class of metal-dependent BSHs (mBSHs) broadly distributed in the gut microbiome. These metalloenzymes have a distinct active site architecture from the canonical Ntn superfamily of cysteine hydolase BSHs (cBSHs) and are selective for taurine-conjugated BAAs. The discovery of this heretofore unappreciated class of BSHs overturns the paradigm that this important, conserved biochemical activity of the gut microbiota is provided exclusively by canonical cysteine hydrolases, greatly expands the known landscape of bile acid metabolism, and reveals a previously unrecognized link connecting host-microbiota bile acid co-metabolism with microbial taurine utilization pathways.