Clathrin is an Intrinsic Driver of Membrane Fission
Clathrin is an Intrinsic Driver of Membrane Fission
Bouzos, N.; Foley, S. L.; Potamianos, A.; Jacobs, C. O.; Johnson, M. E.; Zeno, W. F.
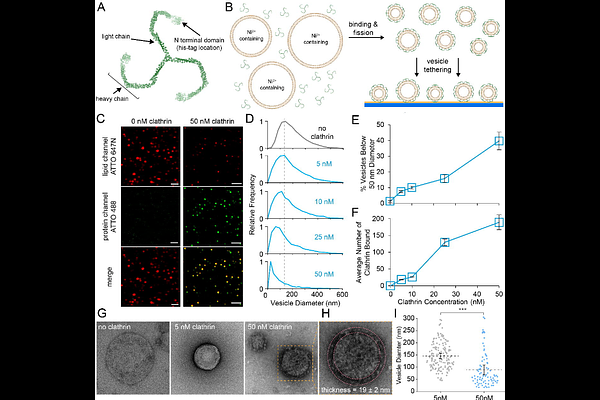
AbstractThe extent to which clathrin directly drives membrane curvature during endocytosis has remained a central, longstanding question in the field of endocytosis. Using a synthetic reconstitution system that recruits clathrin to lipid membranes independent of adaptor proteins, we demonstrate that clathrin alone can induce membrane fission and that its capacity to do so is governed by the mechanical properties of the lattice. Counterintuitively, conditions that strengthen lattice assembly suppress fission, whereas weakened assembly enhances fission despite reduced membrane association. Meso-scale Brownian dynamics reveal that assembly dependent changes in triskelion geometry and lattice stiffness dictate curvature generation, corroborating these findings. These principles are further extended when clathrin is recruited by adaptor proteins amphiphyin1 or epsin1, with clathrin either enhancing or restricting fission based on adaptor specific tuning of lattice mechanics. Lastly, perturbations to clathrin assembly in live cells shift endocytic pit dynamics, with Ca2+; and EGTA producing opposing effects on pit lifetime and productive events consistent with modulation of the membrane fission barrier. Together, these results identify protein and lattice mechanics, not simply bound protein density, as the key determinant of clathrin's ability to remodel membranes, elucidating its biophysical impact on vesicle formation during endocytosis.