The Translatome of Senescent Cells Revealed by Ribosome Profiling
The Translatome of Senescent Cells Revealed by Ribosome Profiling
Kalekar, R. L.; Kelsey, M. M. G.; Sedivy, J. M.
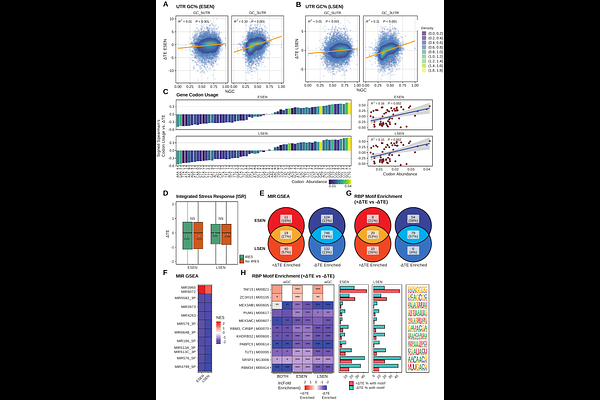
AbstractCellular senescence drives aging-related tissue dysfunction in part through the senescence-associated secretory phenotype (SASP), an inflammatory secretome linked to retrotransposable element (RTE) derepression. Transcriptomic and proteomic approaches have characterized the senescent program extensively, but mRNA abundance does not predict protein output well, and limited proteomic depth constrains the detection of low-abundance SASP factors and RTE-derived proteins. To bridge this gap, we used AHARibo, a metabolic labeling-based method that selectively enriches mRNAs associated with actively elongating ribosomes, to generate translatome profiles in human fibroblasts across proliferating, early senescent, and late senescent states. Comparison of total and ribosome-associated mRNA pools reveals marked translational uncoupling in early senescence: transcriptomic changes explain only 34% of translatomic variance, compared to 70% in late senescence, indicating that early senescence is substantially shaped by post-transcriptional regulation. Key senescence programs are actively regulated at the translational level: cell cycle and extracellular matrix remodeling genes are translationally suppressed and enhanced, respectively, while inflammatory SASP components are translationally depleted in early senescence - a depletion relieved in late senescence. Translationally depleted SASP genes are enriched for binding motifs of the ZFP36 family (ZFP36, ZFP36L1, ZFP36L2), implicating these RNA-binding proteins in the post-transcriptional gating of inflammatory signaling. More broadly, translational efficiency is associated with 3'UTR GC content and codon optimality, and translationally depleted mRNAs are enriched for numerous RBP and microRNA target motifs. Finally, we detect robust, locus-resolved translation of evolutionarily young LINE-1 retrotransposons, identifying full-length elements with stage-specific translational activity. Together, these findings establish translational control as a pervasive regulatory layer shaping the senescent phenotype.