Target RNA abundance controls the collateral activity of RfxCas13d in human cells and zebrafish embryos
Target RNA abundance controls the collateral activity of RfxCas13d in human cells and zebrafish embryos
Chen, H.; Hu, W.; Impicciche, V.; Singh, G. J.; King, J.; Shembrey, C.; Rawat, P.; Casan, J. M. L.; Boskovic, S.; Paterson, S.; Zhao, W.; Lewin, S. R.; Johnstone, R. W.; Hogan, B. M.; Vervoort, S. J.; Trapani, J. A.; Okuda, K. S.; Fareh, M.
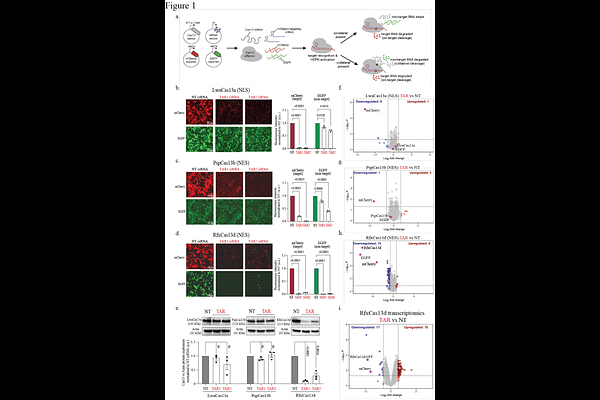
AbstractCollateral RNA cleavage by CRISPR-Cas13 effectors presents a critical obstacle to their application in biological research and therapeutics, yet the molecular determinants of this activity remain poorly understood. Here, we systematically investigate the collateral activity of various Cas13 variants in human cells in vitro and zebrafish embryos in vivo. Among these nucleases, RfxCas13d displays robust collateral activity that is highly dependent on target abundance. Targeting moderately expressed RNAs activates only a limited subset of RfxCas13d molecules, resulting in selective degradation of ectopically expressed transcripts, while endogenous RNAs remain largely protected. This selectivity indicates higher accessibility to exogenous RNAs through spatial proximity and temporal colocalization with activated RfxCas13d nuclease domains. In contrast, the recognition of highly abundant RNA targets drives simultaneous activation of a large fraction of cellular RfxCas13d, leading to widespread collateral cleavage of cellular RNAs, disruption of proteome homeostasis, and consequent cell toxicity and developmental defects in zebrafish embryos. Notably, transgenic zebrafish with target RNA expression restricted to endothelial or neuronal cell lineages exhibit localized collateral activity, leading to tissue-specific developmental abnormalities and motility deficits. These findings reveal that RfxCas13d collateral activity is threshold-dependent, with abundant target RNA acting as a molecular switch for widespread collateral RNA degradation. This work underscores the need for careful consideration of target abundance when deploying RfxCas13d, and highlights PspCas13b as an alternative for RNA silencing free of collateral activity