Terazosin drives sex-dependent adrenergic-bioenergetic reprogramming to restore network function in Alzheimer's disease
Terazosin drives sex-dependent adrenergic-bioenergetic reprogramming to restore network function in Alzheimer's disease
Pattanashetty, S. G.; Serrano, P. A.; Rockwell, P.; Xie, L.; Figueiredo-Pereira, M. E.
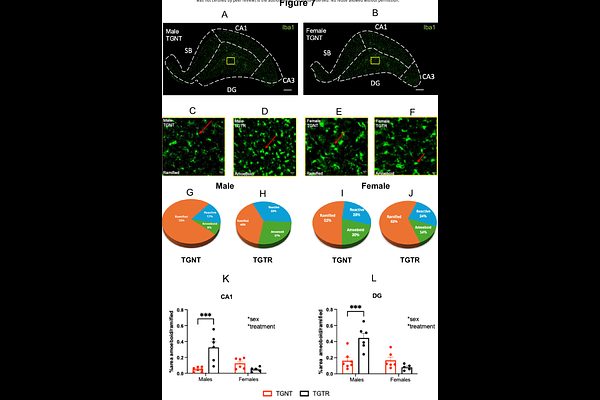
AbstractAlzheimer's disease (AD) has long been defined by amyloid-{beta} plaques and hyperphosphorylated tau, yet disease-modifying therapies remain critically limited. Growing evidence reframes AD as a system-level failure driven by early dysregulation of synaptic, metabolic, and neuroimmune pathways, preceding overt protein aggregation and originating in selectively vulnerable circuits, including the locus coeruleus (LC)-hippocampal noradrenergic axis. This complexity underscores the need for therapeutic strategies that engage the disease at a network level, early in its trajectory. To this end, using a machine learning-based systems pharmacology framework for drug repurposing applied to human AD transcriptomic datasets, we identified terazosin (TZ) as a candidate predicted to reverse AD-associated molecular signatures. TZ is an FDA-approved 1-adrenergic receptor antagonist and phosphoglycerate kinase-1 activator. It was administered chronically via the diet (0.5 mg/kg bw/day) to male and female TgF344-AD rats and wild-type littermates from 5 to 11 months of age, preceding overt pathology. Bulk hippocampal RNA sequencing revealed sex-specific transcriptional remodeling in transgenic rats, strongly conserved with human AD datasets. Male TgF344-AD rats exhibited suppression of synaptic and transcriptional maintenance pathways with concurrent activation of metabolic, proteostatic, extracellular matrix, and vascular stress responses; females showed suppression of survival and vascular structural signaling alongside heightened DAM-like immune activation, amyloid-associated stress, and cell death programs. TZ reversed these signatures in a sex-dependent manner: males showed enhanced immune surveillance and reduced proteostasis burden, while females showed reinforcement of synaptic, survival, and metabolic pathways. TgF344-AD rats displayed selective LC-derived hippocampal noradrenergic axonopathy without global neuronal loss. TZ preserved fiber integrity preferentially in females and partially reversed LC vulnerability-associated transcriptional signatures in both sexes. TZ also reduced amyloid-{beta} plaque burden in both sexes, attenuated hyperphosphorylated tau exclusively in females, and induced microglial morphological shifts in males. Finally, TZ restored wild-type spatial learning in transgenic animals, with females appearing to derive the greater cognitive benefit. Together, these findings demonstrate that TZ induced systems-level reprogramming of AD-relevant molecular pathways and preserved vulnerable noradrenergic circuitry in a sex-dependent manner. Moreover, TZ rescued spatial cognition in transgenic rats, with cognitive gains seemingly more pronounced in females. These results support adrenergic-bioenergetic modulation as a translational strategy for early-stage AD and underscore the necessity of sex as a biological variable in disease-modifying treatment development.