Force-modulated structural landscape of the catch bonding F-actin crosslinker α-actinin-4
Force-modulated structural landscape of the catch bonding F-actin crosslinker α-actinin-4
Chin, A. C.; Mukadum, F.; Reynolds, M. J.; Hocky, G. M.; Alushin, G. M.
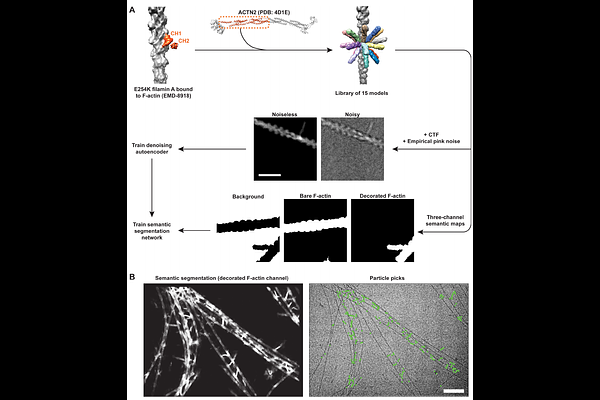
AbstractCatch bonds, noncovalent supramolecular interactions whose lifetimes are increased by force, are ubiquitous in mechanical signaling pathways. The structural mechanisms of catch-bonding proteins remain unclear, hampering efforts to decipher how they are dysregulated in disease and exploit them therapeutically. The crosslinker -actinin-4 (ACTN4) forms catch bonds with actin filaments (F-actin) to support the function of kidney podocytes, and its force-insensitive K255E variant causes autosomal dominant focal segmental glomerulosclerosis (FSGS). Using cryo-electron microscopy (cryo-EM), we find that wild-type ACTN4 engages F-actin in two modes, which biochemical experiments and molecular dynamics simulations assign as strong- and weak-binding states, while K255E ACTN4 only populates the strong binding state. By implementing a cryo-EM platform for applying tension across crosslinker-F-actin interfaces using myosin motors, we find that force promotes a weak-to-strong binding transition for wild-type ACTN4, consistent with a two-state catch bond model. Beyond providing mechanistic insight into how the K255E mutation disrupts ACTN4 F-actin catch-bonding in FSGS, this approach enables structural dissection of force-sensitive actin-binding proteins.