Impaired mitochondrial stress signaling mediates bone loss in male mice in the absence of BNIP3.
Impaired mitochondrial stress signaling mediates bone loss in male mice in the absence of BNIP3.
Tian, L.; Van Berlo, V.; Karthik, V.; passarelli, J. P.; DeMambro, V. E.; Mudjgiwa, P.; Vary, C. P.; Guntur, A. R.
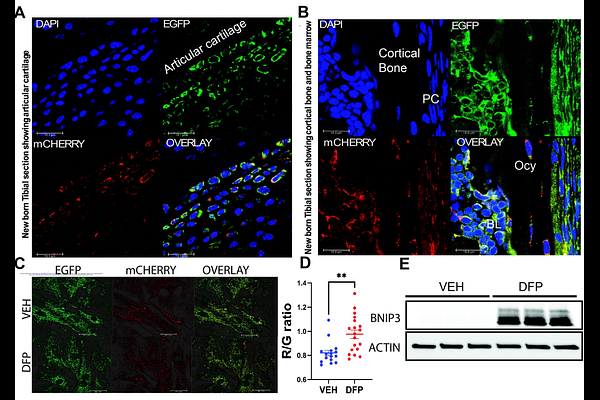
AbstractOsteoblasts generate bone by secreting collagen and mineralizing it in response to various signaling cues. We have previously shown that the majority of ATP generated by differentiated osteoblasts in response to glucose is through glycolysis in contrast to undifferentiated cells that are more dependent on oxidative phosphorylation. To confirm our previous findings, metabolomics was performed for unlabeled polar metabolites, revealing elevated glycolytic metabolites at the later stages of differentiation. Krebs cycle (TCA cycle) metabolites were also changed confirming metabolic rerouting with differentiation. We hypothesized that an increase in mitophagy shifts ATP generation towards glycolysis resulting in the observed bioenergetic and metabolic changes. Utilizing calvarial osteoblasts isolated from a mitophagy reporter mouse model (MitoQC), an increase in mitophagy and the mitophagy receptor, Bnip3, was observed with osteoblast differentiation. Knockdown of Bnip3 in osteoblasts inhibited differentiation and mineralization arising from impaired mitochondrial function. In vivo, male Bnip3 null mice exhibited a significant decrease in osteoblast numbers resulting in lower bone mass. Mechanistically we identified decreased fusion and increased fission factors, impaired stress signaling and increased proapoptotic factors in the absence of Bnip3. These data demonstrate for the first time that BNIP3 expression and mitophagy during osteoblast differentiation are necessary for relieving mitochondrial stress to maintain optimal bone mass.