Cortical inhibitory potentiation reverses maladaptive amygdala plasticity after noise-induced hearing loss
Cortical inhibitory potentiation reverses maladaptive amygdala plasticity after noise-induced hearing loss
Awwad, B.; Polley, D. B.
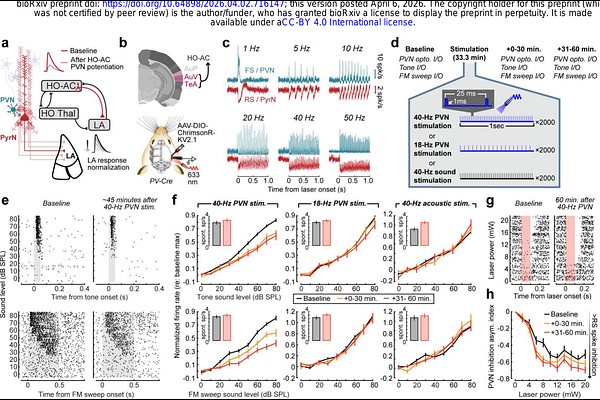
AbstractAcross sensory modalities, peripheral deafferentation can induce maladaptive central plasticity that amplifies sensory responses while distorting affective processing, rendering innocuous stimuli unpleasant, intrusive, or even painful. Here, we examined the affective dimension of compensatory plasticity in a mouse model of hyperacusis by introducing noise-induced focal cochlear lesions and tracking maladaptive plasticity in the lateral amygdala (LA), a key limbic hub for sensory-affective valuation. Whereas control mice habituated to neutral sounds, mice with noise-induced hearing loss (NIHL) showed sustained LA hyperresponsivity and abnormally strong temporal coupling between calcium transients and pupil dilations, an autonomic index of arousal. NIHL also disrupted discriminative auditory threat learning, producing poorly selective, non-extinguishing enhancement of LA responses and freezing to both threatening and non-threatening sounds. We posited that potentiating inhibition in the higher-order auditory cortex (HO-AC), a prominent source of auditory sensory input to the LA, could reinstate normal affective sound processing after NIHL. Brief bouts of 40-Hz optogenetic activation of HO-AC parvalbumin-expressing inhibitory neurons (PVNs) durably reversed LA sensitization, normalized pupil-indexed arousal dynamics, and reinstated discriminative auditory threat memory. These findings identify cortical inhibitory potentiation as a strategy for reversing neural, autonomic, and behavioral signatures of distorted affective sound processing after peripheral sensory injury.