Spatially Organized IGF1 mTOR Signaling Controls Human Forebrain Progenitor Fate Through Coordinated Transcriptional and Translational Programs
Spatially Organized IGF1 mTOR Signaling Controls Human Forebrain Progenitor Fate Through Coordinated Transcriptional and Translational Programs
Xu, J.; Lissit, K.; Huo, D.; Eacker, S. M.; Zhang, S.; Yang, A.; Huang, Y.; Dawson, T.; Dawson, V. L.
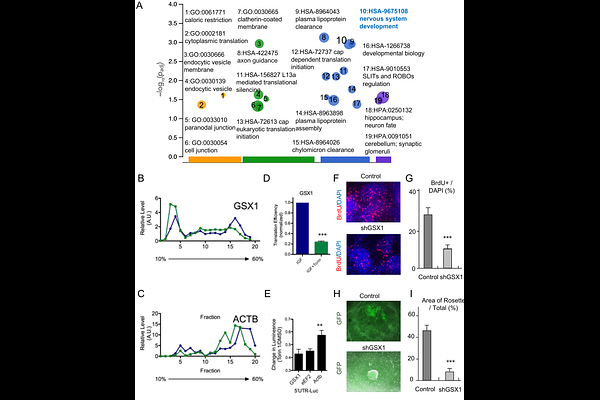
AbstractThe specification and maintenance of human forebrain neural progenitor cells (NPCs) depend on both intrinsic gene networks and spatially localized niche signals, but the interplay between these cues remains incompletely understood. Here, we identify a spatially organized, paracrine IGF1 signaling architecture that regulates human FOXG1 positive NPCs through multilayered transcriptional and translational control. Using a pluripotent stem cell derived forebrain model, we show that FOXG1 positive NPCs express IGF1 receptors but lack endogenous IGF1, instead depending on neighboring epithelial-like domains that secrete IGF1. IGF1 promotes progenitor proliferation, clonal expansion, and vertical tissue growth by activating PI3K AKT mTOR and MEK ERK pathways. Ribosome profiling and 5-UTR reporter assays reveal that mTOR signaling selectively enhances translation of neurodevelopmental and biosynthetic transcripts including GSX1, a ventral fate determinant implicated in interneuron specification and autism. These findings uncover a human-specific regulatory mechanism in which spatially restricted IGF1 mTOR signaling integrates niche signals with translational output to support progenitor identity, biosynthetic capacity, and developmental resilience.