Structural Basis of Electron Transfer by the Human Nitric Oxide Synthase Holoenzyme Complex
Structural Basis of Electron Transfer by the Human Nitric Oxide Synthase Holoenzyme Complex
Lee, K.; Martinez-Ramos, C.; Pospiech, T. H.; Tse, E.; Lau, M.; Osawa, Y.; Southworth, D. R.
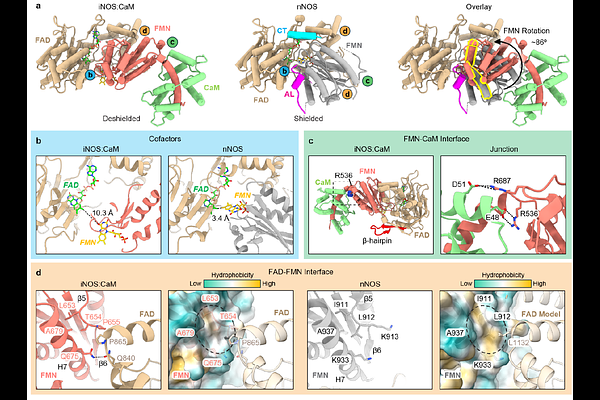
AbstractMammalian nitric oxide synthase (NOS) generates nitric oxide (NO), an essential signaling molecule in neurotransmission, inflammation, and cardiovascular regulation. NOS dysregulation contributes to neurodegeneration, septic shock, and ischemia. The structural basis of NOS function remains unclear, likely due to its dynamic, multi-component architecture. Catalysis requires electron transfer across five cofactors, with the oxygenase (Oxy) domain converting L-arginine to NO after receiving electrons from the reductase (Red) domain via calmodulin (CaM) activation. Here, we report cryo-electron microscopy structures of the human inducible NOS:CaM holoenzyme, revealing a previously unresolved architecture in which the Red:CaM arm spans the Oxy dimer in an apparent active state for cross-monomer electron transfer. The FMN subdomain is rotated ~90{degrees} relative to the Red core, positioning its cofactor ~9 [A] from the heme, adjacent to a conserved aromatic residue within a narrow Oxy domain tunnel. Red--Oxy flexibility in the active state may facilitate electron shuttling among cofactors, defining how NOS cofactors are transiently aligned to enable catalysis.