PI(4,5)P2-dependence of GABAA receptor channel function revealed by optogenetic manipulation of a binding site
PI(4,5)P2-dependence of GABAA receptor channel function revealed by optogenetic manipulation of a binding site
Mori-Kreiner, R.; Andriani, R. T.; Strasdeit, T.; Zhou, J.; Miyashita, N.; Yonezawa, Y.; Kawai, T.; Kloecker, N.; Okamura, Y.
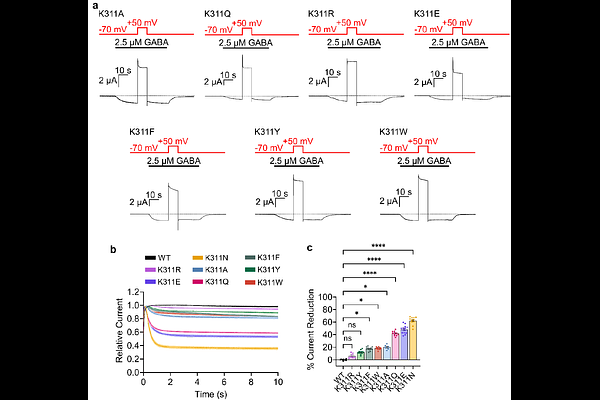
AbstractIonotropic GABAA receptors (GABAARs) mediate fast inhibitory neurotransmission in mammalian brains. While recent structural studies have identified that phosphatidylinositol 4,5-bisphosphate [PI(4,5)P2], a well-established regulator of numerous ion channels, binds to the 1 subunits of GABAARs, the functional relevance of this binding has remained elusive. Here, we combine electrophysiology, molecular dynamics simulations, and a recently developed caged lysine technology to define the role of PI(4,5)P2 in GABAARs. We show that GABAARs are insensitive to acute PI(4,5)P2 depletions by voltage-sensing phosphatase, but sensitivity is conferred by neutralizing the K311 binding site, indicating high-affinity binding. Caging of K311 by use of genetic code expansion recapitulated phenotypes of K311 mutant, conferring sensitivity to PI(4,5)P2 depletion, whereas uncaging restored insensitivity. Furthermore, caging K311 revealed decelerated activation, which then can be accelerated by uncaging. Additionally, PI(4,5)P2-dependence extends to glycine receptors, suggesting PI(4,5)P2 is an important endogenous phospholipid modulator of inhibitory receptor channels.